Introduction
Comorbidity is common among patients with lymphoid cancer (LC) and may limit treatment options. Often, comorbidity is accompanied by medication leading to polypharmacy in multimorbid patients. Whereas comorbidity is difficult to eliminate, discontinuing inappropriate medication may offer an attractive modifiable risk.
Aim
To investigate the effect of polypharmacy and comorbidity on overall survival (OS) in patients with LC.
Methods
We retrieved data on all Danish patients registered with chronic lymphocytic leukemia (CLL) from the Danish CLL Register since 2008 and with mantle cell lymphoma (MCL), diffuse large B-cell lymphoma (DLBCL), and follicular lymphoma (FL) from the Danish Lymphoma Register since 2005. All patients were diagnosed before June 2019; those with more than one LC diagnosis were excluded. Baseline data were used to calculate international prognostic indices (IPI). International Classification of Diseases (ICD10) diagnosis codes were retrieved from the Danish Patient Register, and Charlson comorbidity index (CCI) scores were calculated (Quan et al., Am J Epidemiol, 2011) at time of LC diagnosis. All patients were given a hematological CCI score of 2. Information on medication prescribed within the year up until LC diagnosis was retrieved from the Danish Prescription Register, and the number of drug classes (using the third ATC level, e.g. C03) was used to define polypharmacy as at least 5 drug classes. OS was calculated from time of diagnosis until death or end of follow-up. Multivariable analyses were equally adjusted for CCI, polypharmacy, and appropriate IPI.
Results
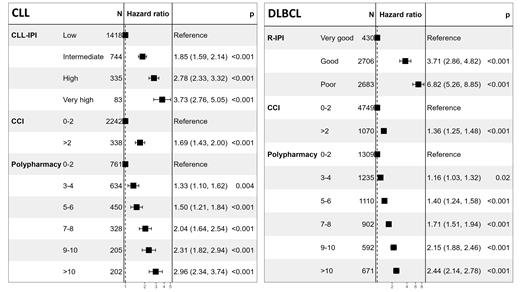
We identified 15,314 patients with LC including 5155 with CLL, 953 with MCL, 6182 with DLBCL, and 3024 with FL. The median age was 69 years (IQR 60;76). CLL-IPI and R-IPI are provided in the Figure. MIPI was low, intermediate, and high in 137 (15%), 297 (32%), and 493 (53%) patients, respectively, and FLIPI2 was low, intermediate, and high in 391 (16%), 1469 (62%), and 518 (22%), respectively. The median CCI score was 2 (including the hematological CCI score of 2, IQR 2;2). Most common comorbidities included chronic pulmonary disease (5%), congestive heart failure (CHF; 3%), rheumatic disease (RD; 3%), and metastatic cancer (MC; 2%). MC (3% vs 1-2%), RD (4% vs 2%), and AIDS (0.3% vs 0.0%) were significantly more prevalent in DLBCL as compared with other LCs ( P≤0.00054).
Patients had most frequently been prescribed antibacterial (J01; 50%), analgesic (N02; 40%), renin-angiotensin system (C09; 33%), antithrombotic (B01; 32%), lipid modifying (C10; 30%), anti-inflammatory/rheumatic (M01; 29%), dyspepsia related (A02; 27%), and diuretic (C03; 27%) drugs. Antibacterials (54% vs 46-51%), analgesics (46% vs 33-36%), dyspepsia related (32% vs 22-28%), anti-inflammatory/antirheumatic (33% vs 24-28%), and systemic corticosteroid (H02; 13% vs 6-8%) drugs were being prescribed significantly more often in patients with DLBCL as compared with those with other LCs ( P<0.0001).
In multivariable analyses adjusted for disease specific IPIs and CCI (2 vs >2), polypharmacy (<5 vs ≥5) was an independent prognostic marker of OS in CLL (hazard ratio [HR] 1.7, 95% confidence interval [CI]; 1.5;2.0), MCL (HR 1.4, 95% CI 1.2;1.7), DLBCL (HR 1.6, 95% CI 1.5;1.8), and FL (HR 1.8, 95% CI 1.6;2.1; P<0.0001). Using polypharmacy as a continuous variable (0-2 vs 3-4 vs 5-6 vs 7-8 vs 9-10 vs >10), we further demonstrated a dose-response effect on OS in CLL, DLBCL (Figure), MCL, and FL (data not shown). Similar to CLL and DLBCL, FL with >2 drug classes were significantly associated with shorter OS, whereas in MCL, polypharmacy groups were small ( n≤262) and only significantly associated with shorter OS for groups with ≥5 drug classes.
Conclusion
Polypharmacy is an important prognostic marker of OS independent of the CCI and IPIs in CLL, MCL, DLBCL, and FL. Polypharmacy is most likely a result of an underlying comorbidity, still informing the prognosis further than register based CCI scores. Further studies will seek to clarify the extent of medication that may be considered for discontinuation in newly diagnosed patients. Moreover, these results indicate that consistent collection of data on both comorbidity and polypharmacy at baseline in clinical trials may improve subgroup analyses and clinical translation of results. This is particularly important when extrapolating clinical trial results to ineligible populations.
Disclosures
Brieghel:Octapharma: Other: Travel grant. Brown:Gilead: Other: Advisory Board; Roche: Other: Advisory Board. Niemann:Carsten Niemann has received research funding and/or consultancy fees from AstraZeneca, Janssen, AbbVie, Beigene, Genmab, CSL Behring, Octapharma, Takeda, and Novo Nordisk Foundation.: Consultancy, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal